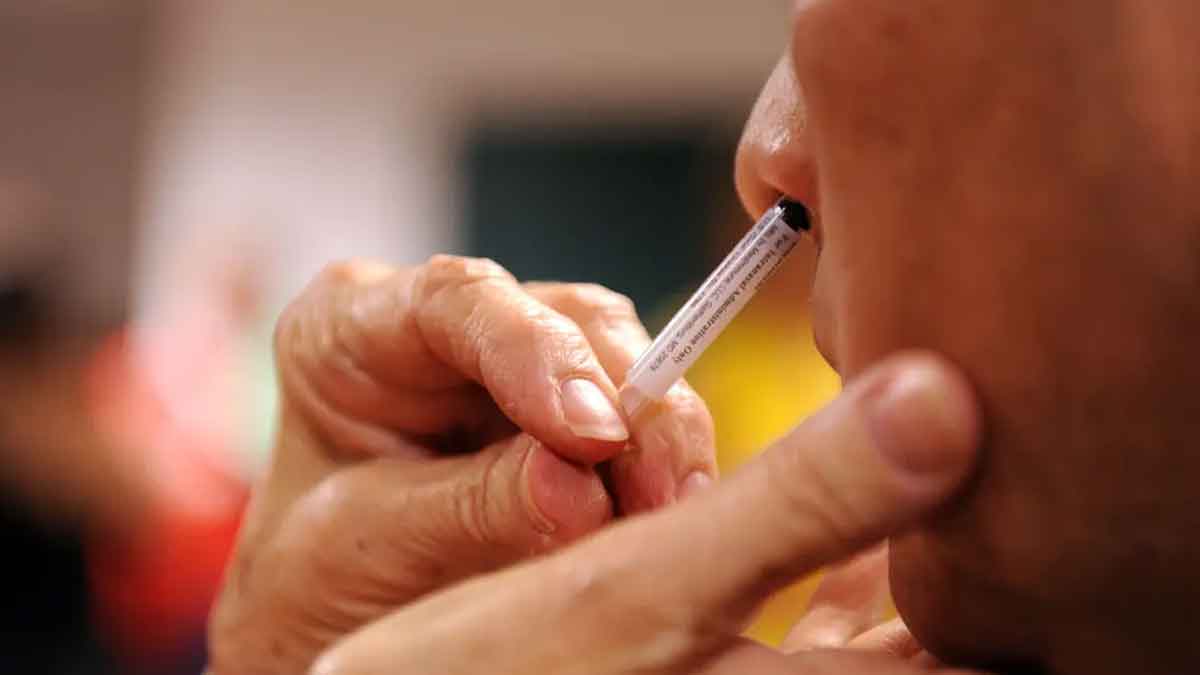
CanSino Biologics Inc. of Tianjin developed the needle-free, inhaled Covid-19 vaccine, and China became the first country to approve it. This caused the company’s shares to rise as much as 14.5% in Hong Kong.
According to a corporate announcement to the Hong Kong Stock Exchange, China’s National Medical Products Administration has authorized CanSino’s Ad5-nCoV for use as a booster vaccine in an emergency situation.
CanSino Biologics of China reported that the country’s drug regulator had given the Covid-19 vaccine it had just created approval for emergency use as a booster, potentially helping its business.
The vaccine is a new version of CanSino’s one-shot Covid drug, which underwent human testing for the first time in the world in March 2020. Since its introduction in China, the vaccine has been used in Pakistan, Malaysia, Mexico and hungry as well. According to CanSino, the inhaled formulation can increase protection without intramuscular injection by stimulating cellular immunity and inducing mucosal immunity.
If the inhaled Covid-19 vaccine is later purchased and deployed by pertinent government entities, the approval “will have a beneficial influence on the company’s performance,” according to CanSino.
Read more: Covid-19 positivity ratio surges to 4.61% as 650 more infected in 24 hours
However, the business issued a cautionary statement, saying that alternative vaccines in China that have already received government approval or are through clinical trials will pose it with strong competition.
Livzon Pharmaceutical Group reported that China has authorized the Covid-19 vaccine, one of just two new anti-disease treatments the government had approved in more than a year, for emergency use as a booster.
Cansino also stated that it was unclear when its inhaled Covid-19 vaccine will be able to reach the market because more administrative clearances are still required, and sales would be contingent on the Covid-19 situation both domestically and internationally as well as China’s vaccination rate.
Covid outbreaks have recently increased in China. Shenzhen, a major tech hub in the south, implemented a weekend lockdown on most of the city. While Chengdu, a major city of 21 million people in the southwest, did the same.
CanSino’s initial one-shot vaccination was found to be 91% effective against severe disease and 66% effective for preventing Covid-19 symptoms, but it lags behind Sinovac Biotech Ltd. and state-owned Sinopharm Group Co. in use outside of China. The majority of the 770 million doses China has delivered to the rest of the world are distributed by those two companies.
In comparison to the 1,988 new cases reported the day before, mainland China reported 1,848 new coronavirus cases for September 3. These cases included both symptomatic and asymptomatic infections.